Prolonged sleep deprivation alters synapses in the brain, but this isn't always entirely negative—it can act as a cleanup mechanism in some cases, though chronic cases heighten risks. Studies indicate that it boosts astrocytic phagocytosis activity, leading to the "eating" of synapses, particularly large and heavily used ones.
Key Points:
- Research confirms that short-term sleep deprivation (6-8 hours) raises astrocytic phagocytosis to 8.4%, compared to 5.7% during normal sleep—this appears as the brain clearing fatigued synapses.
- Chronic sleep deprivation (4-5 days) pushes phagocytosis to 13.5% and activates microglia, which may contribute to synaptic losses and a predisposition to neurodegenerative diseases like Alzheimer's, though not fully proven.
- Sleep deprivation increases synaptic density, but this might be a stress response that disrupts plasticity over time.
- Some studies suggest sleep "resets" synapses, while deprivation disrupts neuronal connections, leading to attention and memory issues.
Basis of the Research
The 2017 study published in The Journal of Neuroscience (by Michele Bellesi and colleagues) was conducted on mice. They compared groups under sleep, wakefulness, short-term, and chronic sleep deprivation. Results show that sleep deprivation enhances astrocytic phagocytosis of synaptic components (especially presynaptic parts). This could be beneficial for shedding excess load, but in chronic cases, microglia join in and pose potential harm. The study measured MERTK gene expression and lipid peroxidation, confirming oxidative stress.
Potential Risks
While sleep deprivation may increase synaptic density (e.g., in the hippocampus), it weakens memory and learning long-term. Recent studies (2021) reveal changes in ribosome-associated transcripts that disrupt synaptic plasticity. In humans, this could lead to attention deficits and neurodegeneration.
Recommendations
Prioritizing sleep is essential—even a short nap (45 minutes) can reset synapses. If you have sleep issues, consult a doctor, as it impacts brain health.
Effects of Prolonged Sleep Deprivation on Brain Synapses
The brain navigates a constant influx of information each day, relying on sleep to restore itself. During sleep, it strengthens synapses, prunes unnecessary connections, and optimizes neural networks. However, prolonged sleep deprivation disrupts this balance, with synapses—the junctions between neurons—bearing the brunt. According to studies in The Journal of Neuroscience, sleep loss activates glial cells in the brain, resulting in the "eating" of synapses. This article delves into the scientific findings, mechanisms, and potential consequences.
Sleep and Brain Plasticity
Sleep serves as a critical recovery process for the brain. It reinforces synapses, eliminates redundant links, and refines neuronal circuitry. The synaptic homeostasis hypothesis posits that wakefulness boosts synapses and their strength, while sleep scales them down—a process called "downscaling." Deprivation upends this equilibrium. In the 2017 study by Bellesi et al., mice under sleep deprivation showed heightened astrocytic phagocytosis, where glial cells engulf synaptic parts. This might aid in clearing overload initially, but chronic deprivation could turn detrimental.
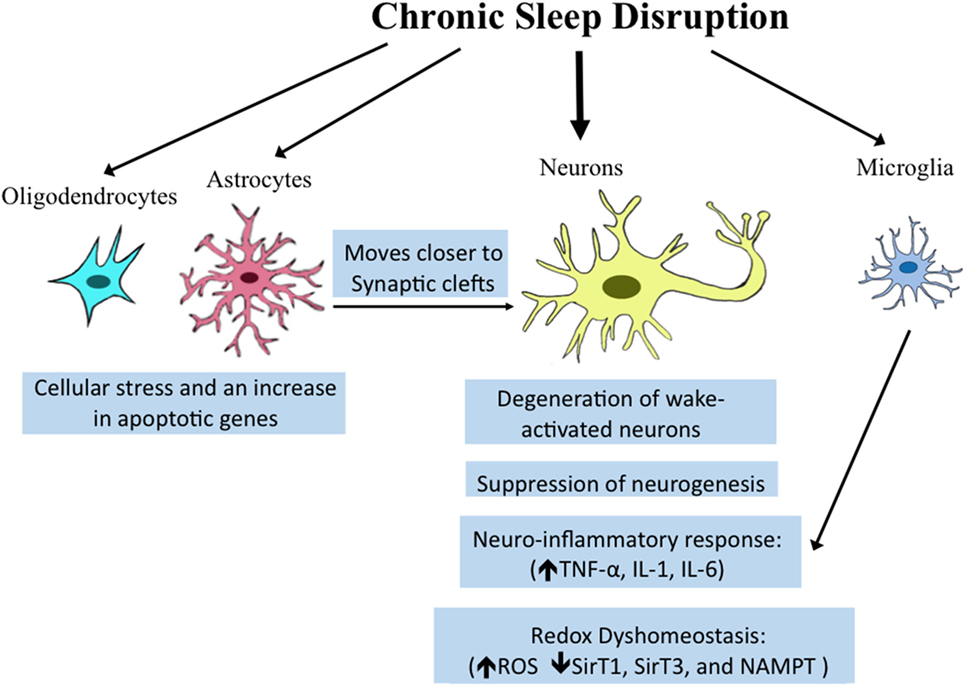 This diagram illustrates the effects of chronic sleep disruption on various brain cells: oligodendrocytes experience cellular stress; astrocytes move closer to synaptic clefts; neurons face degeneration and neuro-inflammatory responses.
This diagram illustrates the effects of chronic sleep disruption on various brain cells: oligodendrocytes experience cellular stress; astrocytes move closer to synaptic clefts; neurons face degeneration and neuro-inflammatory responses.
Methodology and Key Findings
The research utilized mouse models. Four groups were examined: normal sleep (S), spontaneous wake (W), short sleep deprivation (SD, 6-8 hours), and chronic sleep restriction (CSR, 4-5 days). Serial block-face scanning electron microscopy (SBEM) analyzed synapses and astrocytes in the frontal cortex. Outcomes:
- Astrocytic Phagocytosis: In normal sleep, 5.7% of synapses undergo phagocytosis. This rises to 8.4% in short deprivation and 13.5% in chronic cases. Primarily presynaptic components (75%) and large synapses are targeted. This ties to MERTK receptor expression, which surges 30-fold in deprivation, alongside elevated lipid peroxidation (oxidative stress).
- Microglial Role: Only chronic deprivation activates microglia. Process length shortens, ramification decreases, and VGLUT-1 element phagocytosis jumps 28%. The complement system (C3) intensifies, but CSF inflammation markers remain stable, indicating a "primed" state.
Additional studies (de Vivo et al., 2019) found increased synaptic density in the hippocampus during deprivation, but a 18% drop in axon-spine interface. A 2021 investigation highlights ribosomal transcript shifts that impair synaptic plasticity and circadian mechanisms. More recent work (2023) identifies reduced pleiotrophin (PTN) levels in deprived mice, leading to hippocampal cell death via specific pathways.
| Study | Sleep Deprivation Type | Synaptic Effect | Active Cells | Potential Risk |
|---|---|---|---|---|
| Bellesi et al. (2017) | Short (6-8 hours) | Phagocytosis +46% | Astrocytes | Oxidative stress |
| Bellesi et al. (2017) | Chronic (4-5 days) | Phagocytosis +138% | Astrocytes + Microglia | Neurodegeneration risk |
| de Vivo et al. (2019) | Short | Increased synaptic density | - | Memory weakening |
| Delorme et al. (2021) | Chronic | Ribosomal changes | Neurons | Plasticity disruption |
| Havekes et al. (2016) | Short | Dendritic loss in CA1 | - | Memory deficit |
| Qing et al. (2023) | Chronic | Decreased PTN levels | - | Neuronal death in hippocampus |
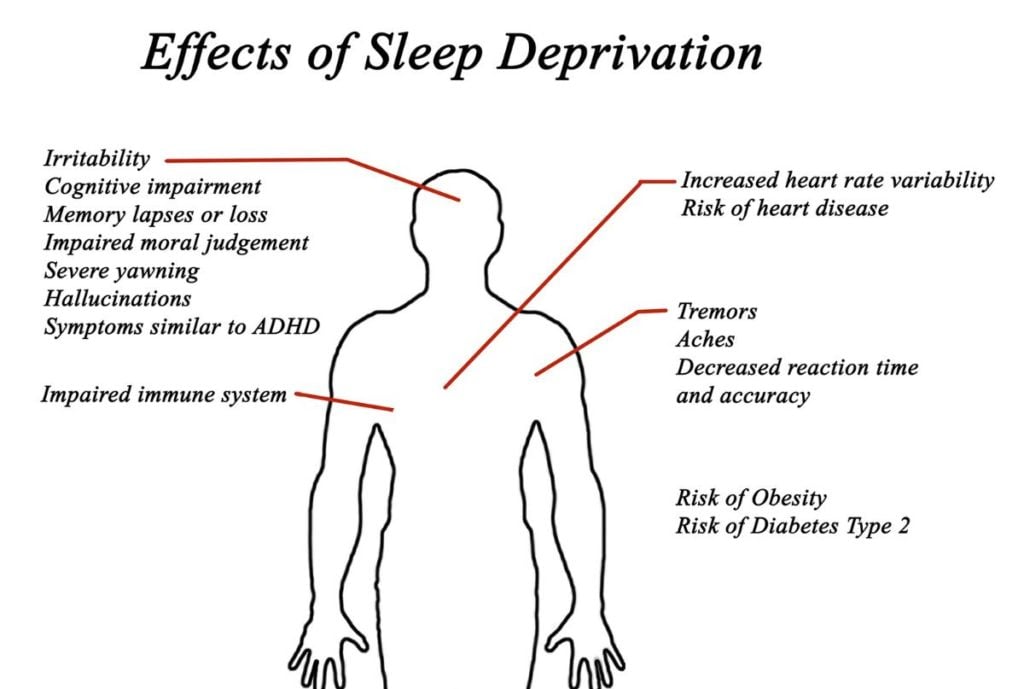
This illustration shows body-wide effects of sleep deprivation: cognitive impairments, memory loss, and immune system weakening.
Mechanisms: Why Does the Brain "Eat Itself"?
Sleep deprivation heightens synaptic activity, generating oxidative stress. Astrocytes detect damaged synapses via phosphatidylserine and phagocytose them. This can be advantageous—clearing worn synapses—but chronic scenarios recruit microglia, raising inflammation risks. In humans, this may accumulate beta-amyloid, heightening Alzheimer's susceptibility. However, some sources caution that the "brain eating itself" phrasing oversimplifies; it's more about excessive pruning that could contribute to disorders over time, not immediate self-cannibalism.
Emerging findings (2025) link deprivation to attention lapses via neurovascular and CSF dynamics. Brief sleep can reset synapses, but deprivation bypasses this opportunity. Broader effects include impaired memory consolidation, increased calorie cravings, and weight gain due to frontal cortex deactivation and mesolimbic hypersensitivity.
Conclusions
Prolonged sleep deprivation shifts the brain into an intensified cleanup mode, but excess can result in synaptic losses and neuronal damage. Studies emphasize chronic forms as more hazardous, though human evidence remains indirect. Future neuroimaging could track these processes. Protecting sleep is the best safeguard; minimum 7 hours daily supports memory, metabolism, and mental health. While short-term deprivation might aid synaptic housekeeping, sustained loss risks neurodegeneration—balancing views note this as a nuanced process, not alarmist self-destruction.
Key Citations
- Sleep Loss Promotes Astrocytic Phagocytosis and Microglial Activation in Mouse Cerebral Cortex
- The brain starts to eat itself after chronic sleep deprivation
- The Neuroprotective Aspects of Sleep
- Poor sleep can eat away at your brain
- Neural consequences of chronic sleep disruption
- The Mammalian Brain Starts Eating Itself When It Doesn't Get Enough Sleep
- Fact Check: Is Sleep Deprivation Making Your Brain 'Eat Itself'?
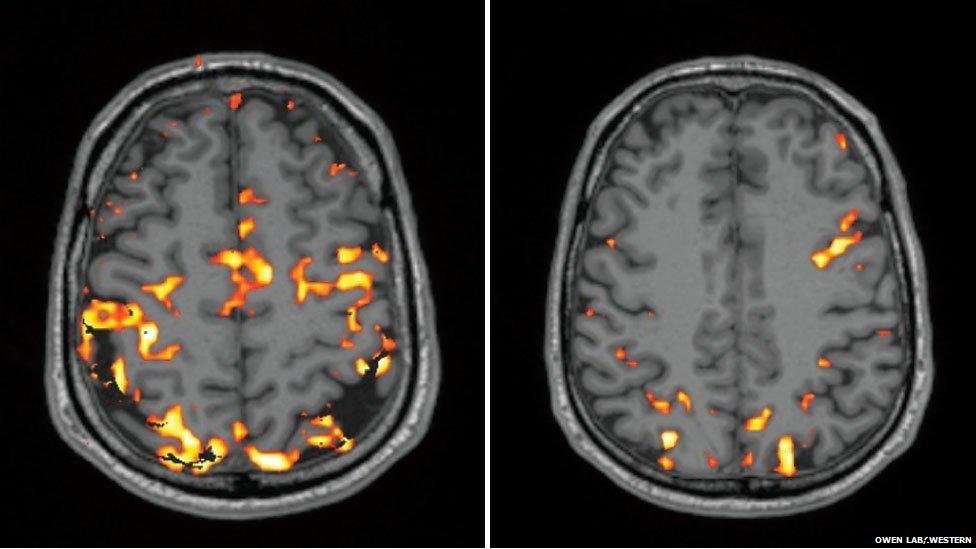
- The sleep-deprived human brain
- How sleep deprivation can harm the brain
- The Brain May 'Eat Itself' When Sleep-Deprived
- Sleep deprivation causes memory deficits by negatively impacting neuronal connectivity in hippocampal area CA1